Xin Dong, Tianqi Chen, Jing Liang, Lei Wang, Huajie Wu, Zhijin Xu, Junhua Luo, Lina Li*
Shaonan Liu, Shuixing Dai*, Minghua Huang
Chin. J. Struct. Chem., 2024, 43: 100277. DOI: 10.1016/j.cjsc.2024.100277
June 15, 2024
ABSTRACT
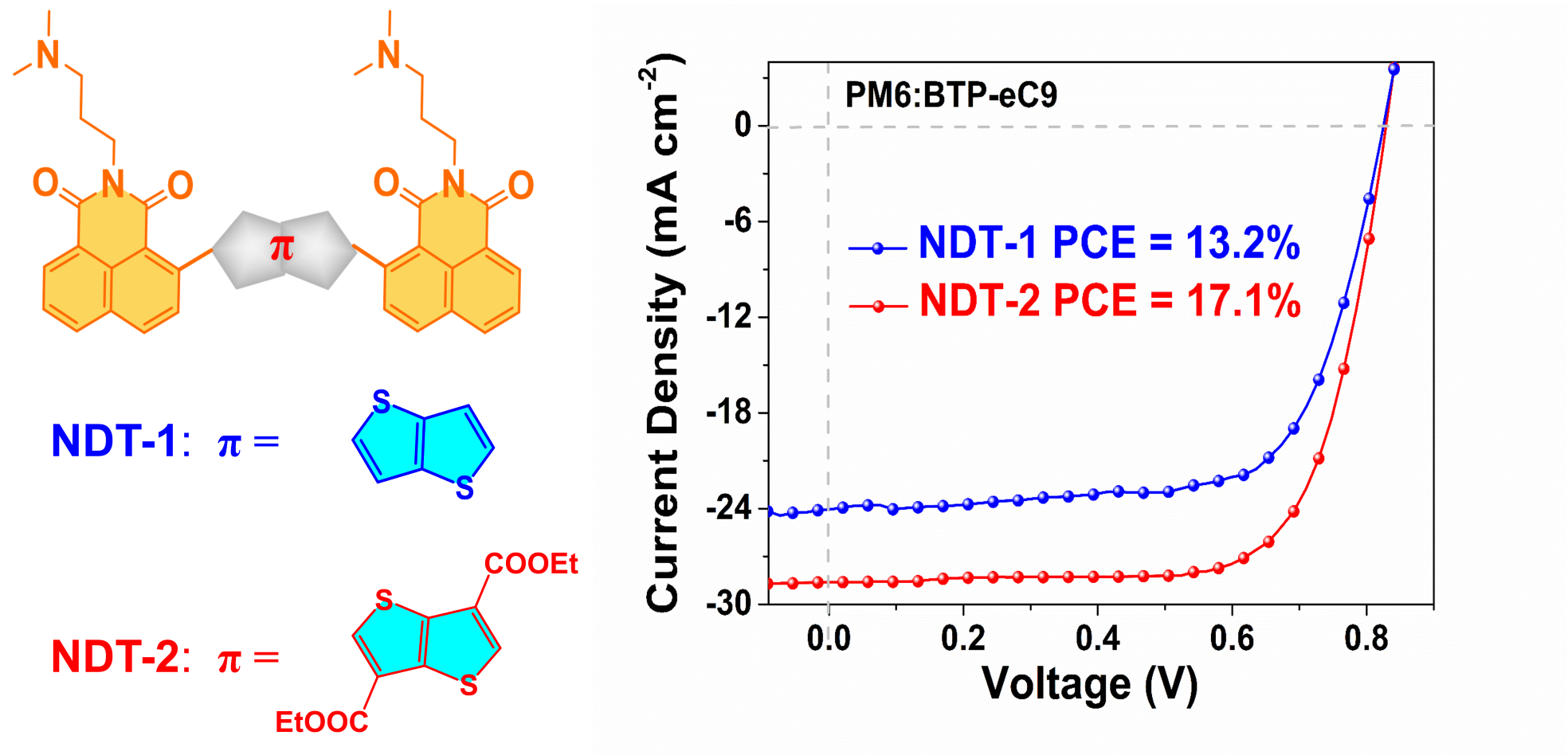
In summary, this study reported the synthesis of two naphthalene imide small molecules, namely NDT-1 and NDT-2, which were successfully used as ETMs in OSCs. The introduction of ester functional groups at the π-bridge units in NDT-2 resulted in (1) an enlarged molecular dihedral angle, improved alcohol solubility, enhanced film conductivity and film-forming quality; (2) suppression of charge recombination, enhancement of electron mobility and reduction of series resistance, leading to a significant increase in JSC; (3) almost no effect on VOC and FF. Ultimately, OSC devices based on PM6:BTP-eC9 with NDT-2 as the ETM achieved a remarkable PCE of 17.1% (a VOC of 0.839 V, a JSC of 28.2 mA cm-2 and FF of 72.1%), demonstrating superior photovoltaic performance compared to NDT-1 (PCE = 13.2%). This work discovered that the ester-modified NI molecules exhibit excellent alcohol solubility and film-forming ability, providing valuable insights for the development of NI molecules as efficient ETMs used in high-efficient OSCs