Just Accepted
Just Accepted Articles have been posted online after technical editing and typesetting for immediate view. The final edited version with page numbers will appear in the Current Issue soon.
Submit a Manuscript

Accelerated water dissociation kinetics by electron-rich hydrous RuO2 for efficient alkaline hydrogen evolution reaction
Jingmei Min#, Hao Huang#, Xueyan Wu, Yan Lv, Jiaxin Li, Hongmei Zhang, Fei Song, Jixi Guo*, Dianzeng Jia*
https://doi.org/10.1016/j.cjsc.2026.100980
Electron transfer; Electron-rich; Hydrous RuO2; Water dissociation; Alkaline HER
ABSTRACT
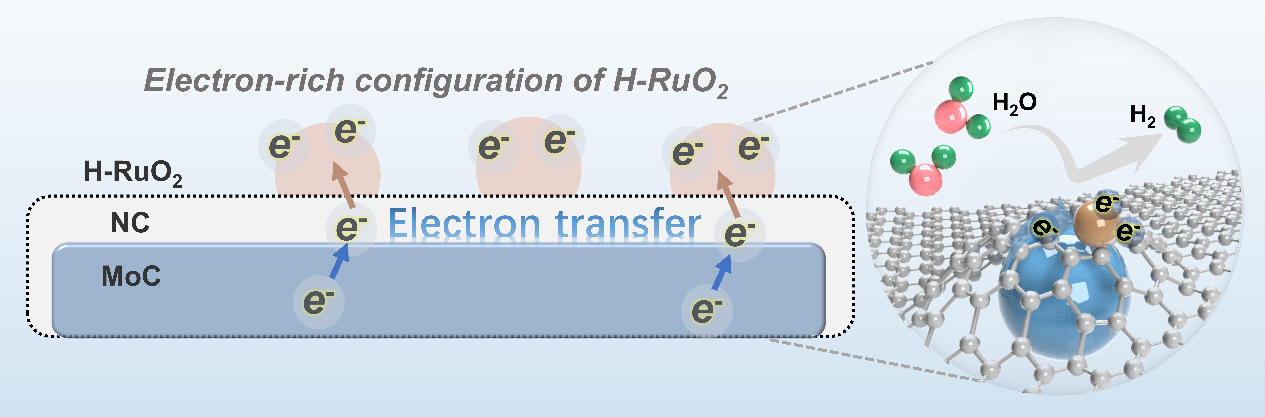
Ruthenium-based catalysts have attracted extensive attention as promising alternatives to platinum-based materials in the alkaline hydrogen evolution reaction (HER). However, the oxidized state of Ru suffers from electron deficiency, which hinders its HER performance and complicates the understanding of the underlying catalytic mechanisms. Herein, hydrous RuO2 (H-RuO2) supported on an N-doped carbon (NC)-coated MoC (MoC@NC) is developed to electron-enrich the H-RuO2 for effectively enhancing the alkaline HER performance. The prepared catalyst exhibits remarkable alkaline HER performance, with a low overpotential of 27 mV at 10 mA cm-2 and a small Tafel slope of 24.8 mV dec-1 in 1.0 M KOH, surpassing most Ru-based electrocatalysts and commercial Pt/C. Experimental results and density functional theory (DFT) calculations reveal that the strong interfacial interactions between H-RuO2 and MoC@NC promote the formation of electron-rich H-RuO2, elevating its d-band center towards the Fermi level, which enhances *OH intermediate adsorption and facilitates the OH–H bond cleavage for proton production and lowers the energy barrier of H2O dissociation, leading to improved HER activity. These findings provide an effective strategy for the rational design of Ru-based electrocatalysts for sustainable hydrogen production.