Just Accepted
Just Accepted Articles have been posted online after technical editing and typesetting for immediate view. The final edited version with page numbers will appear in the Current Issue soon.
Submit a Manuscript

Tuning effective conjugation via B←N orientation: High-mobility n-type isoindigo polymers for organic field-effect transistors
Jingshu Tian, Haofeng Sun, Jiarui Li, Cheng Wang, Kun Yang, Qinqi Zhou, Kai-kai Liu*, Maning Liu*, Haichang Zhang*
https://doi.org/10.1016/j.cjsc.2026.100943
B←N coordination chemistry; n-type conjugated polymers; Organic field-effect transistors (OFETs); Electron mobility; Backbone conformation
ABSTRACT
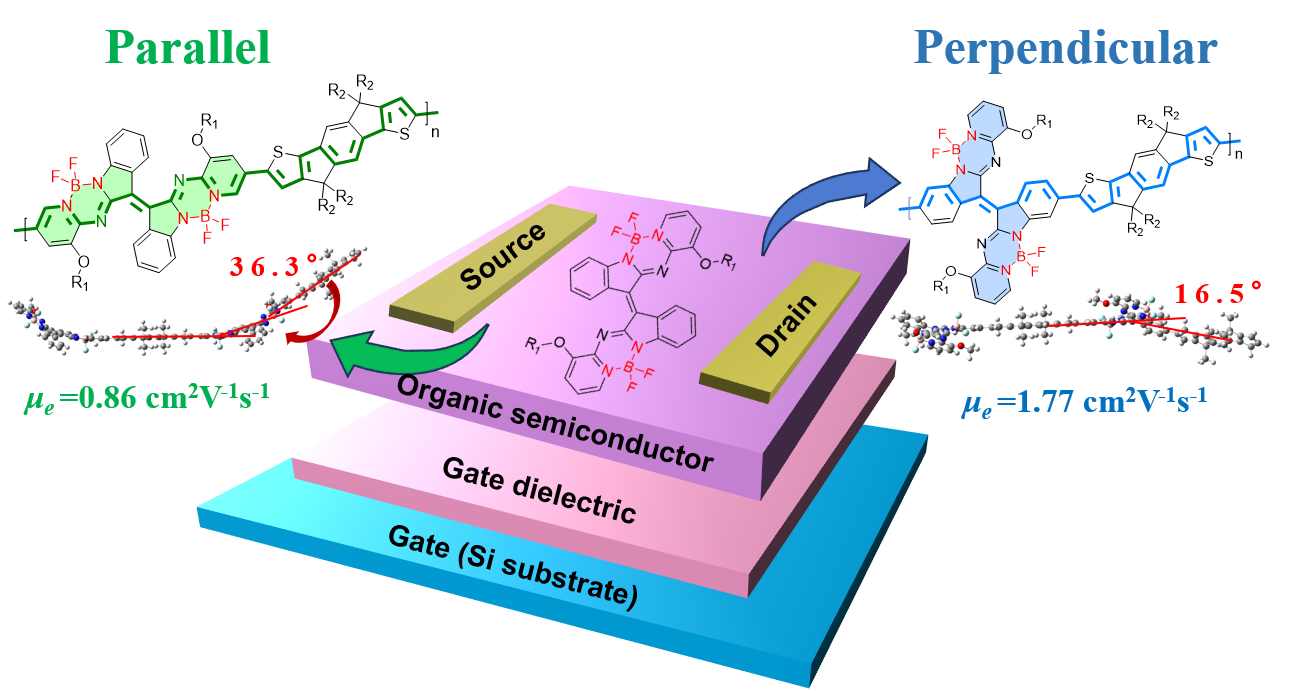
The incorporation of electron-withdrawing B←N coordination units has emerged as an effective strategy to enhance n-type charge transport in conjugated polymers. However, the orientation of these units—whether perpendicular or parallel to the polymer backbone—profoundly influences effective conjugation, molecular packing, and ultimately charge transport properties. To address this structure–property relationship systematically, we designed the IIDG-AB chromophore with precisely controlled polymerization sites, yielding two isomeric polymers: P1 with perpendicular B←N orientation and P2 with parallel orientation. Integrated theoretical and experimental investigations reveal that P1, despite having a shorter geometric conjugation length, achieves a more extended effective π-system, stronger intramolecular charge transfer, tighter solution-phase aggregation, closer π–π stacking, and more delocalized electron excitation. These collective attributes result in a deeper lowest unoccupied molecular orbital level and superior electron transport, with P1 exhibiting an electron mobility of 1.77 cm2 V-1s-1 in organic field-effect transistors—substantially exceeding that of P2 (0.86 cm2 V-1s-1). This work demonstrates that chromophore twist and intrinsic dihedral angles can distort backbone conformation, limit effective conjugation and weaken packing efficiency. We highlight that orienting B←N coordination perpendicular to the backbone is critical, establishing a key design principle for high-performance n-type polymers. The concept of "conformation-driven performance" offers a valuable paradigm for the rational development of organic electronic materials.