Just Accepted
Just Accepted Articles have been posted online after technical editing and typesetting for immediate view. The final edited version with page numbers will appear in the Current Issue soon.
Submit a Manuscript

High-crystalline g-C3N4/BiOIO3 S-scheme heterojunction for efficient photocatalytic CO2 reduction
Zhiyuan Qin#, Haosheng Li#, Song Li, Heng Yang, Juntao Yan, Qiang Ma*, Xiaofang Li*
https://doi.org/10.1016/j.cjsc.2026.100969
BiOIO3; High-crystalline g-C3N4; S-scheme heterojunction; Photocatalysis; CO2 reduction
ABSTRACT
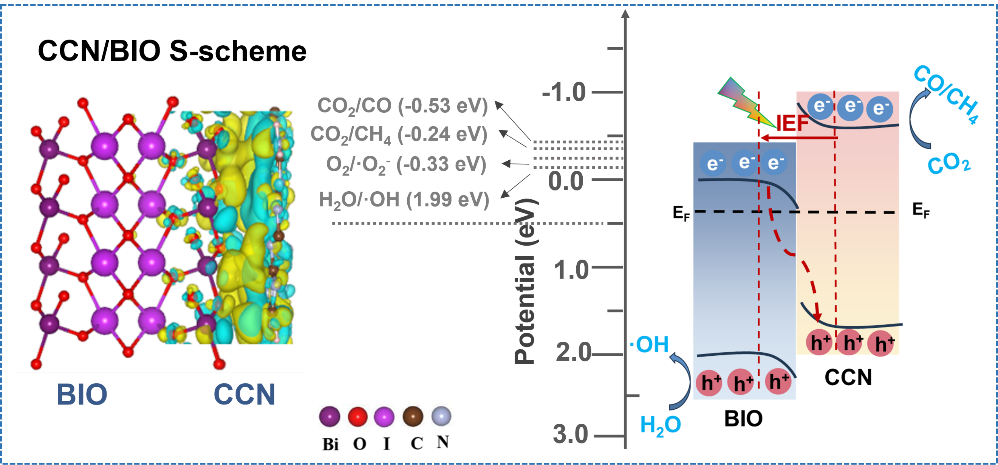
Photocatalytic conversion of CO2 into chemical fuels represents a promising route toward achieving carbon neutrality. However, its practical large-scale deployment is severely hampered, primarily due to high charge-transfer resistance and sluggish kinetics. Herein, a high-crystalline g-C3N4/BiOIO3 (CCN/BIO) S-scheme is constructed through a one-step KI molten salt method, which offers a greener and simpler alternative to conventional systems while enhancing the crystallinity of CCN and enabling the in-situ deposition of BIO. Benefiting from the high lattice matching between the two components, robust N-O-Bi interfacial chemical bonds are formed, serving as efficient channels for charge migration. As a result, the optimized CCN/BIO composite exhibits a markedly enhanced activity of photocatalytic reduction of CO2, with the total yield of CO and CH4 reaching 4.9 and 3.2 times those of pristine BIO and CCN, respectively. Combined density functional theory (DFT) calculations, in-situ irradiated X-ray photoelectron spectroscopy (XPS), and electron paramagnetic resonance (EPR) analyses systematically verify the S-scheme charge transfer pathway, which is synergistically driven by the internal electric field and interfacial chemical bonds. This work provides new insights into the rational design of visible-light-responsive S-scheme heterojunction photocatalysts.